Risks associated with gases: flammable, toxic or asphyxiating
The air we breathe contains oxygen and nitrogen, and natural gas or methane is used in many homes for heating and cooking... Gas is part of our daily environment, but if we're not careful, it can quickly become dangerous.
There are three categories of gas-related hazards:
- fire and/or explosion risks associated with flammable gases such as methane, butane, propane, etc.
- the risk of poisoning from toxic gases such as carbon monoxide (CO), hydrogen sulfide (H₂S), carbon dioxide (CO₂), chlorine (Cl₂), ...
- risks of suffocation or asphyxiation due to lack of oxygen, which can be consumed or displaced by another gas.

Definition and types of gas-related risks

By definition, a gas is a swarm of molecules that move randomly and chaotically, colliding with each other and with their surroundings. Gases fill any available volume and, because of their speed of movement, they mix rapidly throughout the atmosphere into which they are introduced. Gases can be heavier, lighter or of the same density as air. Some are odorous, others odorless. Some are colored. Just because you can't see, smell or touch a gas doesn't mean it isn't there.
The word gas comes from the Latin "chaos", meaning the immense, dark space that existed before the origin of things in mythology. The word was introduced in the 17th century by Flemish chemist Jean-Baptiste Van Helmont to describe a notion of emptiness.
Understand the risks related to flammable gases

Hydrocarbon compounds such as acetylene, ammonia, hydrogen, propane, propylene and methane are all flammable gases. They are also known as combustible gases.
Combustion is a chemical reaction in which oxygen rapidly mixes with another substance, releasing energy in the form of heat or flames.
Combustion can be represented by the fire triangle, in which three factors must come together:
- A source of ignition
- Oxygen
- Fuel in the form of gas or steam
That's why the aim of any fire protection system is to eliminate one of these potentially dangerous elements.

Flammability or explosive limits
The flammability or explosive limits of a gas refer to the minimum and maximum concentrations of the gas necessary to support its combustion in the air (burning or explosion):
- The lower limit is called the LEL (Lower Explosive Limit)
- The upper limit is called the UEL (Upper Explosive Limit)
Below the LEL, the mixture is said to be lean, as there is insufficient gas to produce an explosion. The risk of fire increases as the percentage of flammable gas rises. Above the LEL, the mixture lacks oxygen. The air becomes too rich to burn, reducing the chances of ignition. As a general rule, an increase in pressure, temperature or concentration increases the flammability range.
Typically, industrial plants do not leak gas, or only trace amounts. Gas detectors are used to monitor risks prior to combustion, i.e. to measure gas concentrations between 0% and the lower explosive limit (LEL). When the lower limit is reached, site shutdown or evacuation procedures are initiated. In practice, these measures tend to be taken at concentrations below 50% of the LEL value, to provide a sufficient safety margin.
In confined spaces (enclosed or unventilated environments), concentrations above the LES can sometimes occur. The danger lies in the opening of doors and hatches, as the inflow of air from outside can dilute the gases to form a dangerous combustible mixture. For this reason, some gases are scented to facilitate leak detection.
Autoignition temperature
Above a certain temperature, flammable gases ignite even without a spark or flame. This temperature is called the ignition point. The surface temperature of devices used in hazardous areas must not exceed the ignition point. A maximum surface temperature is therefore indicated on the devices.
Flash point (freezing point in °C)
This is the lowest temperature at which the liquid surface produces sufficient vapors to ignite a small flame.
Vapour density
Elle aide à déterminer l’emplacement du capteur. La densité de gaz/vapeur est comparée à l’air : lorsque l’air = 1,0 alors Densité de vapeur < 1 augmente et Densité de vapeur > 1 chute.
Understand the risks related to toxic gases
Flammable gases and toxic gases are treated separately because they involve different risks, regulations and sensors. However, there are many gases that are both combustible and toxic, meaning that toxic gas detectors have to be approved for hazardous areas.
In the case of toxic substances, the main concern – aside from environmental issues - is the effect of these gases on employees. Inhaling, ingesting or having one of these gases penetrate your skin may be harmful even at very low concentrations. The number of deaths caused by exposure to toxic gases is higher than the number of deaths due to explosions involving flammable gases.
The most common way of measuring the concentration of toxic gases is in parts per million (ppm) and parts per billion (ppb). For example, 1ppm is the equivalent of a room filled with a million balls, 1 of which is red: the red ball represents 1ppm. However, it is essential to measure the exposure time, not just the concentration of gas, because the harmful effects are often caused by regular exposure over a long period of time.
VLEPs
An important notion is that of Occupational Exposure Limit, or OEL. The purpose of the OEL is to prevent irreversible damage to a person’s health caused by the use of one or more hazardous substances. It offers protection against adverse effects on health, but does not cover the risk of explosion. In other words, the goal is to make sure that the exposure levels in the workplace are lower than the acceptable legal limit. These limits are a valuable tool for risk evaluation and management.
To obtain a representative measurement of the degree of contamination of the air inhaled, three elements are required:
- a field study (profile of potential exposures)
- personal surveillance via wearable sensors
- sampling as close as possible to the breathing zone.
The toxicity of the gas in question must absolutely be taken into account. For example, a detector that only measures a weighted average, or one that takes a sample for laboratory analysis, does not protect an employee against a short exposure to a lethal dose of an extremely toxic substance. However, it may be normal to temporarily exceed long-term exposure limit levels in certain parts of a plant without necessarily triggering an alert situation. The optimum detector should therefore be able to monitor both long-term and short-term exposure levels, as well as instantaneous alarm levels.
Exposure times are estimated over eight hours (VLEP-8h) and 15 minutes (VLCT). For some substances, brief exposure is so critical that they have only a short-term exposure limit, which must never be exceeded, even for a very short time. Carcinogenicity, reproductive toxicity, irritation and sensitization are all taken into account when drawing up a proposal for an OEL based on current scientific knowledge.
Understand oxygen-related risks
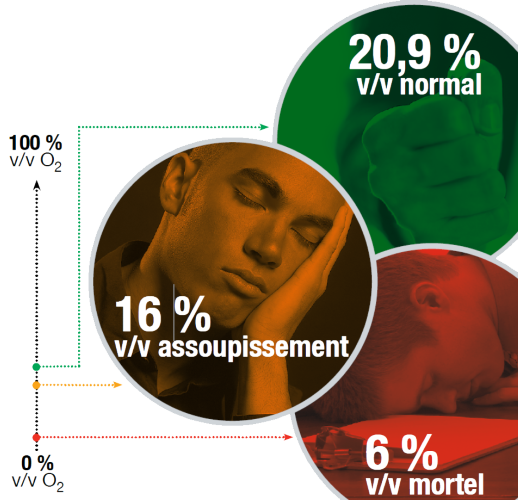
In the case of oxygen deprivation (or asphyxia)
We all need to breathe the oxygen (O₂) in the air in order to live. However, oxygen is just one of the many gases present in the air. Normal ambient air contains an oxygen concentration of 20.9% v/v. When the oxygen level falls below 19.5% v/v, the air is considered oxygen-deficient. Oxygen concentrations below 16% v/v are considered unsafe for humans.
Oxygen depletion can be caused by:
- Displacement
- Combustion
- Chemical reaction
- Bacterial action
In the case of oxygen enrichment
The danger posed by air too rich in oxygen is also often overlooked. Increased O₂ increases the flammability of materials and gases. Above 24%, spontaneous combustions can occur, particularly of clothing.
Oxyacetylene welding combines oxygen and acetylene gas to produce extremely high temperatures. Oxygen-enriched atmospheres can represent a hazard in other environments, such as manufacturing or storage areas for reaction propulsion systems and bleaching products from the paper industry, or water treatment plants.
Sensors must be specifically certified for use in O₂-enriched atmospheres.
Our ranges for gas detection

Portable Gas detector

Fixed Gas Detector




